The New McDonald Criteria: Is Your MS Study Ready?
Discover how the new McDonald criteria use imaging biomarkers such as the Central Vein Sign (CVS) to make earlier, more accurate MS diagnosis
Discover how the Central Vein Sign (CVS) — with 95% sensitivity and 92% specificity — is transforming MS diagnosis and how QMENTA powered the CAVS-MS trial validating CVS as a diagnostic biomarker

MS affects 2.8 million people worldwide, over 80% of which have varying degrees of disability. MS research has shown that the disease not only impacts physical abilities, but also mental health and personal independence.
Patients experience a severe decline in emotional quality of life, suffering from depression or heavy emotional burden; some even have suicidal thoughts. (8)
Despite the urgency of early treatment to slow disease progression and disability, diagnostic delays remain a major problem, affecting more than 50% of patients with MS. (19)
Some patients are misdiagnosed by neurologists (19) not giving them the chance to start the appropriate therapy on time.
An early diagnosis means that early treatment initiation can start to preserve brain tissue and optimize brain health. This can maximize the chances of altering the disease course before further relapses or disability progression occurs. (5)
A diagnosis of MS relies on the integration of clinical, imaging, and laboratory tests. This is summarized in the current iteration of the McDonald criteria (20), whereby diagnosis is based on the principles of dissemination in space (DIS) and dissemination in time (DIT) of clinical attacks and/or central nervous system (CNS) lesions and the exclusion of other diseases mimicking MS. (14)
In simple terms, this means finding MS lesions in different areas of the CNS and demonstrating that they occurred at different times (6), as well as excluding disease that present similar symptoms (12) or may present a similar appearance on magnetic resonance imaging (MRI). (1)
It has been shown that several factors, both disease-related and local, can contribute to the diagnostic delay. In particular, the difficulty of recognizing typical MS disease features, both in images and symptoms, and local factors such as access to MRI. (14)
It is now clear that MRI is crucial for the diagnosis of MS; however, it is not generally very specific, often requiring a diagnosis of exclusion, meaning that other diseases must be excluded before a diagnosis of MS can be confirmed. (17)
In short, MS is a complex condition, difficult to diagnose and easily confused with other diseases. Although the diagnosis integrates clinical, imaging and laboratory tests, none of these are highly specific for MS. The main cause of diagnostic problems is often the misinterpretation of MRI scans. (13)
As a result, the importance of identifying more specific MRI biomarkers for MS is evident.
In this ongoing purpose to improve MS diagnosis and rule out MS-mimicking disorders, a biomarker has recently shown great promise: the central vein sign (CVS). (17)
The CVS is a radiological finding on neuroimaging, indicating the presence of a central vein within a white matter lesion, which is characteristic of MS. (4)
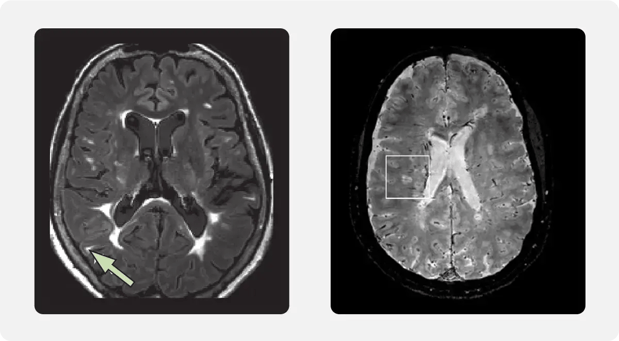
Left image: Hyperintense lesion near the cortex; source: Filippi et al. MRI criteria for the diagnosis of multiple sclerosis: MAGNIMS consensus guidelines, Lancet Neurol 2016:15(3). Right Image: Image of how some MRI sequences can show lesions and CNS vessels simultaneously; source: Gill et al. Emerging imaging and liquid biomarkers in multiple sclerosis, Eur J Immunol 2023:53(8)
What started 13 years ago as an hypothesis by Nikos Evangelou’s team at Nottingham (18) has grown into a recent advancement for the early diagnosis of MS. CVS in MS lesions first emerged in 2011 and was proposed as a potential MS biomarker (7), then in 2016 the North American Imaging in MS Cooperative issued a consensus statement on its presence (15). From 2017 onwards, the scientific community turned its attention to discovering the potential of CVS, both to understand its role in the disease and to refine detection techniques.
In 2021, the CAVS-MS trial (Central Vein Sign: a Diagnostic Biomarker in Multiple Sclerosis) was born, a pioneering effort to validate CVS as a diagnostic biomarker.Clinical trials like CAVS-MS are pivotal in advancing multiple sclerosis research, and QMENTA has played a key role in managing the data infrastructure supporting this groundbreaking research.(11)
CAVS-MS clinical trial was initiated as a collaborative effort to support the MS clinical trials landscape. This imaging trial highlights the potential of innovation and technology, as well as QMENTA’s commitment, to advancing the understanding of MS and so improving patient outcomes.
The trial sees the QMENTA Imaging Hub serve as a centralized repository and infrastructure for all participating parties, 2 institutions and 9 sites: Cleveland Clinic - collaborating with Dr Daniel Ontaneda, Cedars-Sinai in collaboration with Dr Pascal Sati, and then a number of other participating sites: Johns Hopkins University, University of Pennsylvania, St. Michael's Hospital, University of Colorado, University of South California, University of Texas at Austin, University of Vermont, Washington University and Yale University.
At QMENTA, we manage the implementation of the CAVS-MS workflow, which has led to significant improvements in the efficiency and accuracy of clinical data management for CVS research in MS.
QMENTA’s workflow implementation includes:
By automating protocol adherence rules, defining 16 distinct user roles and digitizing and implementing electronic reporting forms (eCRF) with dynamic logic, we simplify and ensure the entire process from data entry to data analysis is seamless and robust.
Our query management system ensures data integrity by flagging inconsistencies for a timely resolution, while our ongoing support throughout the project enables its smooth operation.
This whole process not only optimizes data quality, but also allows researchers to focus on what really matters, advancing CVS research.
CVS-MS: From Emerging Indicator to Diagnostic Biomarker
In 2023, the use of advanced techniques such as FLAIR* allowed researchers to refine the detection of CVS and it was recommended for the first time to be included in the McDonald criteria. (12)
Now, in 2024, the CVS is being considered for inclusion in the diagnostic criteria for MS, representing a potential breakthrough in multiple sclerosis research and diagnosis...as a diagnostic biomarker has been confirmed, with the recent announcement of its incorporation into McDonald's diagnostic criteria (9).
| Criteria | McDonald 2017 | Proposed McDonald 2024 | Goal |
|---|---|---|---|
| MRI Role | MRI remains central for assessing DIS and DIT | Expanded use of advanced MRI techniques, CVS and PRLs may be used for diagnosis | Improve diagnostic detail and accuracy |
| DIS (Dissemination in Space) | MRI scans should show lesions in at least two out of four typical CNS regions | Includes optic nerve as a 5th CNS location. Lesions in at least 4 regions or CSF markers alone are now sufficient for MS diagnosis | Improve accuracy in diagnosing MS |
| DIT (Dissemination in Time) | The presence of new lesions on MRI or positive oligoclonal bands (OCBs) in the CSF supports progression | DIT no longer required if DIS criteria are met. Kappa free light chains added as a substitute for OCBs | Detect MS sooner by using additional biomarkers alongside MRI |
| Neurofilament Light Chain (NFL) | Not used | Likely to be used as a biomarker for nerve damage and progression risk in CIS and RIS | Improve detection of nerve damage and predict MS progression risk |
| Radiologically Isolated Syndrome (RIS) | Not included in diagnostic criteria | In specific cases, RIS can now be diagnosed as MS if certain risk factors and imaging findings are present | Treat RIS patients with higher risk of developing MS more proactively |
| Primary Progressive MS (PPMS) | Diagnosis requires at least one year of progressive disability, supported by MRI or CSF evidence of MS | Faster diagnoses may be possible through advanced imaging and biomarkers | Diagnose PPMS sooner and allow earlier treatment options |
| Differential Diagnosis | Emphasis on excluding conditions that mimic MS | Enhanced imaging techniques, biomarkers, and AI are increasingly helping to differentiate MS from similar diseases | Reduce misdiagnosis by enhancing diagnostic tools and adding AI |
| Monitoring RIS, CIS, Early MS | Routine MRI follow-ups are recommended, with no immediate treatment for RIS or CIS unless symptoms emerge | A more structured approach considers MRI findings, biomarkers, and risk factors to guide treatment decisions for RIS and CIS | Ensure patients with RIS/CIS receive timely monitoring and potential treatment if risks are high |
CVS has emerged as a very promising diagnostic tool for MS. In fact, CVS was found in 73% of lesions in patients with MS, showing a 95% of sensitivity and 92% of specificity.
A cut-off value of 40% of lesions presenting CVS has been demonstrated (3); in other words, if more than 40% of white matter brain lesions show CVS, the likelihood to diagnose MS is very high. The diagnostic accuracy of CVS is reliable with a sensitivity and specificity of 90% and 89%. (3)
In essence, CVS turns out to be highly specific and sensitive for typical MS lesions and as such is of particular value in differentiating MS from other conditions on MRI. (3)
MRI at 3T and higher field strengths are sufficiently clear to be practically useful, whereas lower field magnets (1.5T) demonstrate significant degradation in detection accuracy. (3)
In addition to diagnostic performance, CVS also has practical advantages. Unlike other neuroimaging markers, it is visible to the naked eye with appropriate sequences, and, being a binary marker, simplifies the diagnostic process. This simplicity is particularly important in both typical and atypical MS clinical presentations, where early and accurate diagnosis can lead to a timely therapeutic intervention. (10)
In conclusion, including CVS as a diagnostic criterion has the potential to significantly improve specificity over currently employed methods and makes it an important tool for diagnosis and post-diagnostic management of MS (10). Higher specificity means less room for error and uncertainty, which increases the possibility of having accurate diagnoses at the right time, therefore being able to start the right therapies in a timely manner. Clinical trials like CAVS-MS and ongoing neuroimaging advancements continue to push the boundaries of what’s possible, providing new tools to reduce diagnostic delays and improve patient care.
The Central Vein Sign is a radiological finding on MRI neuroimaging that indicates the presence of a small central vein within a white matter lesion — a feature characteristic of MS pathology. In MS, inflammatory cells infiltrate the surrounding white matter from the central vein, creating a perivenular lesion that can be visualised on susceptibility-sensitive MRI sequences. The CVS was first proposed as a potential MS biomarker in 2013 and has since been validated through multi-site clinical research, culminating in its incorporation into the updated McDonald Criteria for MS diagnosis in 2024.
Studies show that the CVS was found in 73% of lesions in patients with MS. A systematic review and meta-analysis (Castellaro et al., 2020) established diagnostic accuracy with a sensitivity of 90% and specificity of 89% using a cut-off of 40% of lesions presenting CVS. Separately, meta-analyses report sensitivity of 95% and specificity of 92% for CVS in MS lesions. A cut-off value of 40% — meaning more than 40% of white matter brain lesions showing CVS — has been validated as a practical threshold for high-confidence MS diagnosis.
Diagnostic delays affect more than 50% of patients with MS. The primary challenge is that conventional MRI findings used in MS diagnosis — white matter hyperintensities — are non-specific and appear in many conditions including migraine, small vessel disease, and other inflammatory disorders. Without a highly specific imaging biomarker, MS diagnosis often requires a lengthy process of exclusion. The CVS, visible to the naked eye on appropriate MRI sequences and binary in its interpretation, offers a practical specificity boost — reducing the diagnostic ambiguity that drives delays and misdiagnosis.
The CAVS-MS trial (Central Vein Sign: a Diagnostic Biomarker in Multiple Sclerosis; ClinicalTrials.gov: NCT04495556) is a pioneering multi-site clinical study initiated in 2021 to validate CVS as a diagnostic biomarker in a real-world multi-centre, multi-vendor setting. The trial involved 2 lead institutions (Cleveland Clinic with Dr. Daniel Ontaneda; Cedars-Sinai with Dr. Pascal Sati) and 9 additional participating sites including Johns Hopkins, University of Pennsylvania, Yale, and Washington University. QMENTA served as the centralised imaging data management infrastructure, implementing the CVS detection workflow, managing 16 distinct user roles, deploying 23 custom eCRFs, and providing automated protocol adherence checking across all sites.
The CVS requires susceptibility-sensitive MRI sequences, most commonly FLAIR* (a post-processing combination of FLAIR and T2* sequences). MRI at 3 Tesla (3T) and higher field strengths provide sufficient image clarity for practical CVS detection. The evidence shows significant degradation in detection accuracy at lower field strengths of 1.5T. The FLAIR* technique, developed by Sati et al. in 2012, is currently the recommended method for CVS detection in clinical and research settings, providing enhanced contrast between both white matter lesions and cerebral veins compared to standard sequences.
The CVS and PRLs are both susceptibility-sensitive MRI biomarkers incorporated into the 2024 McDonald Criteria, but they reflect different aspects of MS pathology. The CVS indicates the acute perivenular inflammatory origin of an MS lesion, making it valuable for diagnosis — particularly differentiating MS from mimicking conditions. PRLs, by contrast, represent chronic smouldering inflammatory activity at lesion edges (visible as an iron-rich rim), associated with greater disease severity and ongoing demyelination. PRLs are therefore more valuable as a marker of disease progression and as a therapeutic endpoint in clinical trials, while the CVS is primarily a diagnostic specificity tool.
Discover how the new McDonald criteria use imaging biomarkers such as the Central Vein Sign (CVS) to make earlier, more accurate MS diagnosis
Learn how QMENTA simplifies central vein detection in Multiple Sclerosis. Our solution automates protocol adherence, sequence recognition, and white...
It is difficult to diagnose Parkinson’s disease, and the only way to confirm diagnosis is postmortem. The new imaging AI tools could be the answer.